Visceral Lesion Applicator
Visceral Lesion Applicator (VLA)
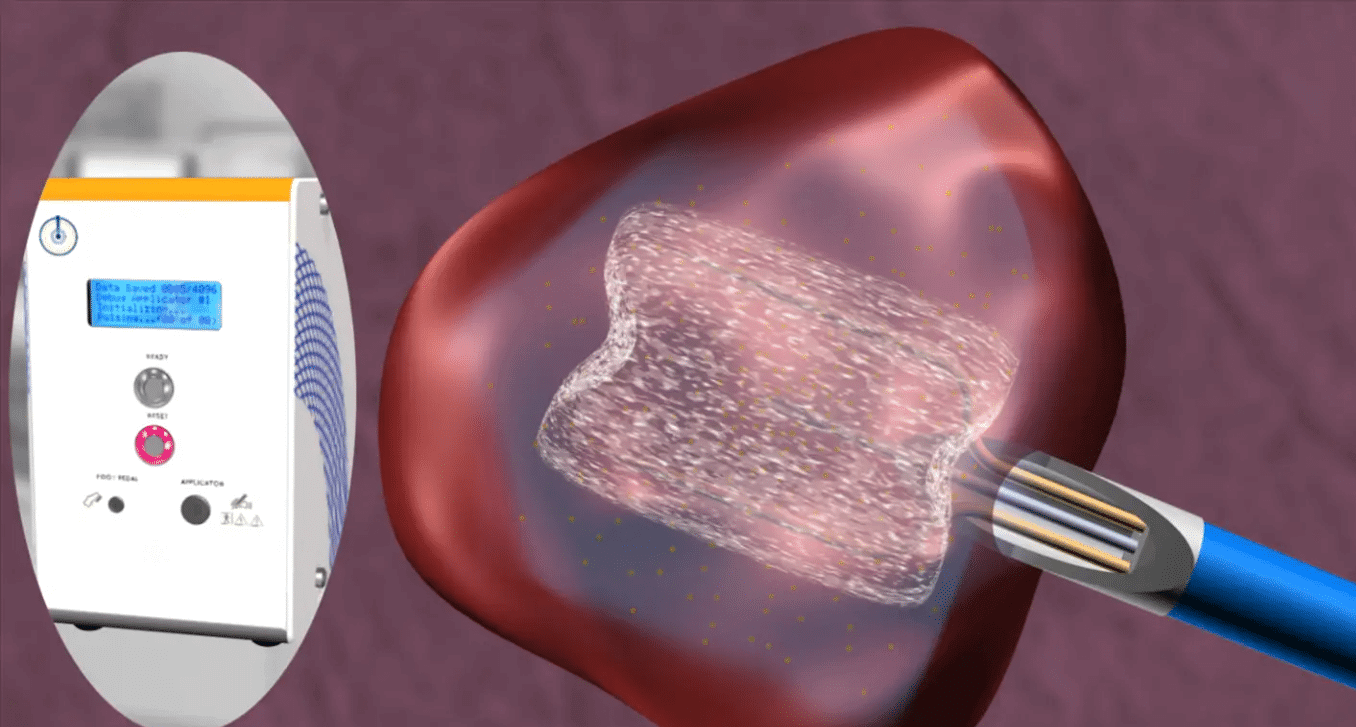
OncoSec’s investigational visceral lesion applicator (VLA) technology is designed to treat non-cutaneous, internal tumors through direct delivery of our lead product candidate, TAVO™, or other immunologically relevant genes, including our SPARK plasmid. Lesions located within organs of the gastrointestinal tract, lung, liver, pancreas and bone can often be difficult to treat successfully with conventional therapies. Currently, there are no FDA-approved intratumoral, direct-injection immunotherapy options for cancers other than unresectable, metastatic melanoma.
This investigational, next-generation intratumoral delivery platform will be comprised of novel access applicators, both rigid and flexible catheter-based, as well as a new reversible, low-voltage electroporation generator (APOLLO).
The applicator options could enable the ability to use a variety of interventional and surgical approaches to access a wide array of lesions, such as primary and metastatic lesions of the lung (flexible catheter-based) and liver (rigid-based). Enabling the delivery of immunotherapy directly to these types of lesions could represent a meaningful advancement in treatment options for patients.
In preclinical feasibility models, we have successfully demonstrated the ability of both applicator types (flexible and rigid) to access target organs. We plan to complete preclinical assessment work for VLA in 2020 and enter human trials in 2021.
VLA Corporate Presentation
OncoSec’s VLA 3D MOA Animation
OncoSec's VLA 3D MOA Animation
Scientific Presentations
SIR 2020 – Can an intratumoral DNA-encoded immunotherapeutic device platform currently used in the management of cutaneous lesions be scaled in size to function in the treatment of visceral tumors through image-guided techniques?
SIO 2020 – Novel Controlled Delivery of Potent Anti-Cancer Immunotherapy Directly to Deep Visceral Lesions
The VLA applicators and APOLLO generator have not been tested in humans.
