COMBINATION APPROACH
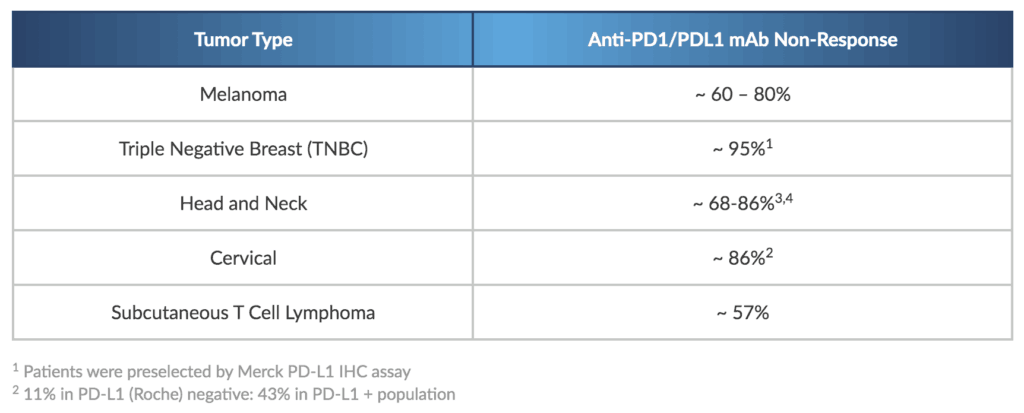
The majority of patients with solid tumors who have been treated with anti-PD-1/PD-L1 therapies do not respond to treatment: this is one of the great challenges in oncology today. OncoSec our investigational platform believes TAVO™ may address this unmet medical need by increasing the proportion of patients who will respond to anti-PD-1 and other checkpoint therapies.
How It’s Designed to Work
Certain cancers can bypass or hide from the immune system by engaging an immune checkpoint called PD-1, which is a protein found on T-cells. PD-1 represents an “off-switch” which cancers can exploit to escape from T-cells, which are critical to the immune system’s anti-tumor attack. New treatments, called checkpoint inhibitors, block these “off-switches” and help keep T-cells in attack mode to fight cancer cells.
Patients who possess “switched-off” anti-tumor CD8+T-cells in their tumors (i.e. increased tumor-infiltrating lymphocytes, or TILs) are most likely to respond to therapy with anti-PD1 or anti-PD-L1 drugs. However, studies show that these TILs – the target cell for T-cell checkpoint agents – are lacking in the majority of patients. As a result, the majority of patients with solid tumors are anti-PD-1 non-responders.
Pre-clinical data indicates that local delivery and expression of TAVO promotes tumor immunogenicity and increases TILs. As a pro-inflammatory cytokine, IL-12 can promote the recruitment of T-cells to the tumor. By driving T-cells or TILs into the tumor microenvironment, TAVO may enhance response to anti-PD-1 and convert anti-PD-1 non-responders to responders.
Additional Resources
The following scientific resources provide additional information about immune checkpoint therapies in cancer immunotherapy.
- Herbst RS, et. al. “Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients.” Nature. 2014 Nov27;515(7528): 563–567. doi:10.1038/nature14011. >> Read More
- Tumeh PC, et. al. “PD-1 blockade induces responses by inhibiting adaptive immune resistance.” Nature. 2014 Nov 27;515(7528):568-71. doi: 10.1038/nature13954. >> Read More
- Pardoll DM. “The blockade of immune checkpoints in cancer immunotherapy.” Nat Rev Cancer. 2012 Mar 22;12(4):252-64. doi: 10.1038/nrc3239. >> Read More
- “Tumor Immunogenicity: The Role of PD-1 and TILs.” Presented by Dr. Robert H. Pierce, MD, Chief Scientific Officer, OncoSec Medical >> See Video
Clinical Pipeline
Our core technology platform continues to advance through clinical trials for various cancers
Technology
Our plasmid DNA delivery platform is designed to boost the body’s immune system to target and attack cancer
Partnerships
We have a track record of establishing, operating and evolving high-performance partnerships globally
